Determination of Palladium Content in Pd/C
1 Sample solution preparation:
2 Experimental equipment and reagents:
3 Instrument conditions
4 Standard solution preparation
5 Standard curve
0.1g sample (precision to 0.0001g) was weighed and placed in a 250mL beaker, 15mL aqua regia (HCl: HNO3=3:1) was added, heated on hot plate. After digestion, it was transferred to a 100mL volumetric flask, rinsed and made up to the volume with deionized water, shaken well. Then 0.25mL sample solution was pipetted into a 100mL volumetric flask, then diluted and made up to the volume with deionized water, shaken well and spared for later use.
2 Experimental equipment and reagents:
AA7000 series atomic absorption spectrophotometer (with Pd hollow cathode lamp, EWAI Inc.)
Temperature-controlled hot plate
Nitric acid (HNO3): excellent grade purity
Hydrochloric acid (HCl): excellent grade purity
Palladium standard solution (National Reference Materials Research Center)
Temperature-controlled hot plate
Nitric acid (HNO3): excellent grade purity
Hydrochloric acid (HCl): excellent grade purity
Palladium standard solution (National Reference Materials Research Center)
3 Instrument conditions
| Parameter | Wavelength (nm) |
Slit width(nm) | Burner height(mm) | Fuel gas flow rate(L/min) | Lamp current(mA) | Flame type |
| Pd | 244.8 | 0.2 | 10 | 1.5 | 2.0 | Air - acetylene |
4 Standard solution preparation
| Element | Concentration(μg/mL) | ||||
| Pd | 0 | 0.5 | 1.0 | 1.5 | 2.0 |
5 Standard curve
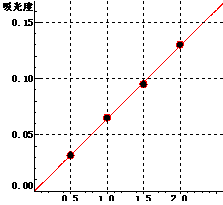 Pd Curve equation:y=0.0646*x-0.0002 Linearity coefficient:0.99969 |