Analysis of volatile components in the anti-acne cosmetic by GC-MS
1 Experiment
1.1 Instrument
GC-MS3200 (EAST & WEST ANALYTICAL INSTRUMENTS, INC); 10mL microinjector;
commercial anti-acne cosmetic
1.2 Analysis conditions
GC:Equity-5 (30m×0.25mm×0.25um) silica capillary column; injection port temperature:280℃; split sampling; sample volume: 0.2mL; split ratio: 60:1; precolumn pressure: 100 kPa; purge rate:2ml/min; column temperature:hold at 80℃ for 1min, ramp up to 280 °C at 10 °C/min and then hold for 10 min.
MS:EI source; ion source temperature: 200℃; electron energy: 70eV; scanning mode:full scan;
scanning range: 25u~400u; interface temperature:280℃; multiplier high voltage:1100V.
2 Results and discussion
1.1 Instrument
GC-MS3200 (EAST & WEST ANALYTICAL INSTRUMENTS, INC); 10mL microinjector;
commercial anti-acne cosmetic
1.2 Analysis conditions
GC:Equity-5 (30m×0.25mm×0.25um) silica capillary column; injection port temperature:280℃; split sampling; sample volume: 0.2mL; split ratio: 60:1; precolumn pressure: 100 kPa; purge rate:2ml/min; column temperature:hold at 80℃ for 1min, ramp up to 280 °C at 10 °C/min and then hold for 10 min.
MS:EI source; ion source temperature: 200℃; electron energy: 70eV; scanning mode:full scan;
scanning range: 25u~400u; interface temperature:280℃; multiplier high voltage:1100V.
2 Results and discussion
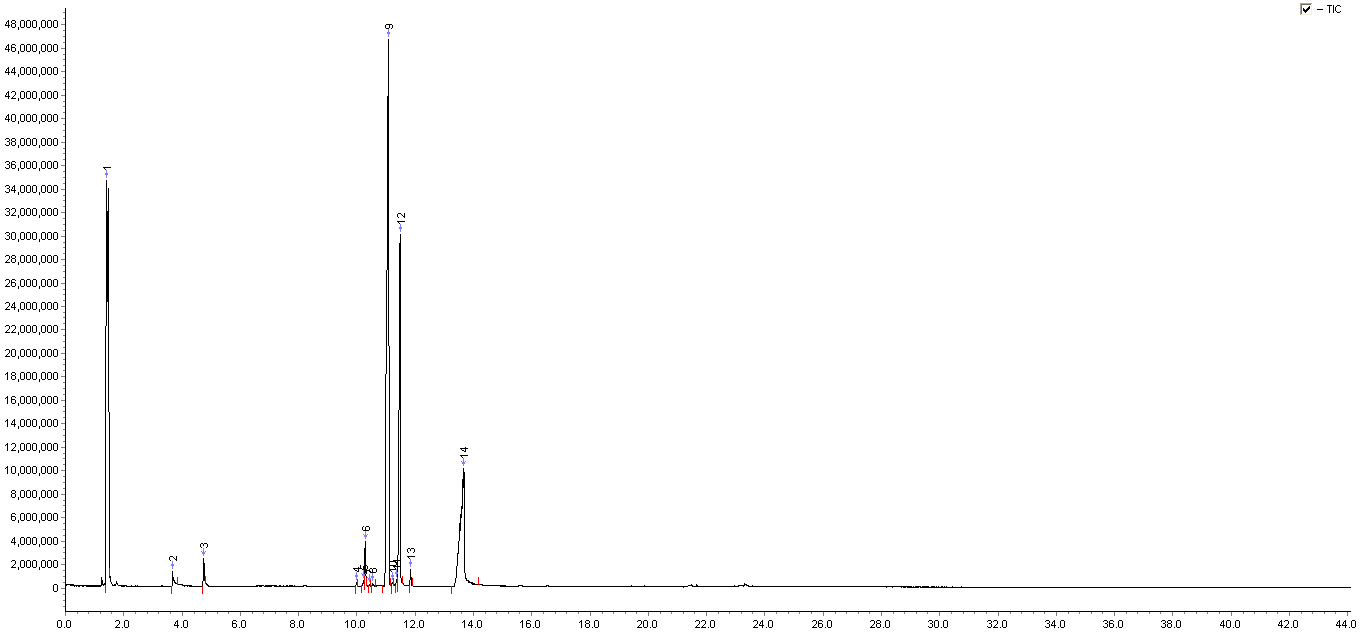
Figure 1 An anti-acne cosmetic’s chromatogram
GC-MS3100 was used to analyze the composition of a commercially available anti-acne cosmetic; Figure 1 shows the full-scan chromatogram of sample. Through NIST standard library search combined with manual analysis, 14 components were detected (see Table 1). The main components are Bicyclo[2.2.1]heptan-2-one, 1,7,7-trimethyl-, (1R)-, menthol, salicylic acid, etc., in which ethanol should be a solvent; most components have a matching degree of more than 80%.
As weakly polar capillary column was used in this experiment, the peak shape of individual components with higher polarity in the sample are not ideal, and if highly polar column is used, the peak shape of polar components would be better.
Table 1 Analysis results of the anti-acne cosmetic
| No. | Retention time/min | Name | Molecular formula | CAS | similarity degree /% |
| 1 | 1.4 1 | Ethanol | C2H6O | 64-17-5 | 93 |
| 2 | 3.67 | Propylene Glycol | C3H8O2 | 57-55-6 | 91 |
| 3 | 4.74 | Propanoic acid, 2-hydroxy-, ethyl ester, (S)- | C5H10O3 | 687-47-8 | 88 |
| 4 | 9.99 | Bicyclo[2.2.1]heptan-2-one, 1,3,3-trimethyl- | C10H16O | 1195-79-5 | 86 |
| 5 | 10.23 | Bicyclo[4.1.0]heptan-3-ol, 3,7,7-trimethyl-, (1S,3R,6R)-(+)- | C10H18O | 4017-79-2 | 77 |
| 6 | 10.29 | Acetaldehyde, (3,3-dimethylcyclohexylidene)-, (E)- | C10H16O | 26532-25-2 | 82 |
| 7 | 10.43 | Bicyclo[2.2.1]heptan-2-ol, 1,5,5-trimethyl- | C10H18O | 6168-62-3 | 72 |
| 8 | 10.54 | Camphor | C10H16O | 76-22-2 | 70 |
| 9 | 11.00 | Bicyclo[2.2.1]heptan-2-one, 1,7,7-trimethyl-, (1R)- | C10H16O | 464-49-3 | 95 |
| 10 | 11.22 | Isoborneol | C10H18O | 124-76-5 | 83 |
| 11 | 11.35 | Borneol | C10H18O | 507-70-0 | 85 |
| 12 | 11.48 | Menthol | C10H20O | 1490-04-6 | 95 |
| 13 | 11.84 | Bicyclo[2.2.1]heptan-2-one, 1,7,7-trimethyl-, (1S)- | C10H16O | 464-48-2 | 83 |
| 14 | 13.67 | Salicylic Acid | C7H6O3 | 69-72-7 | 91 |
3 Some component’s mass spectra
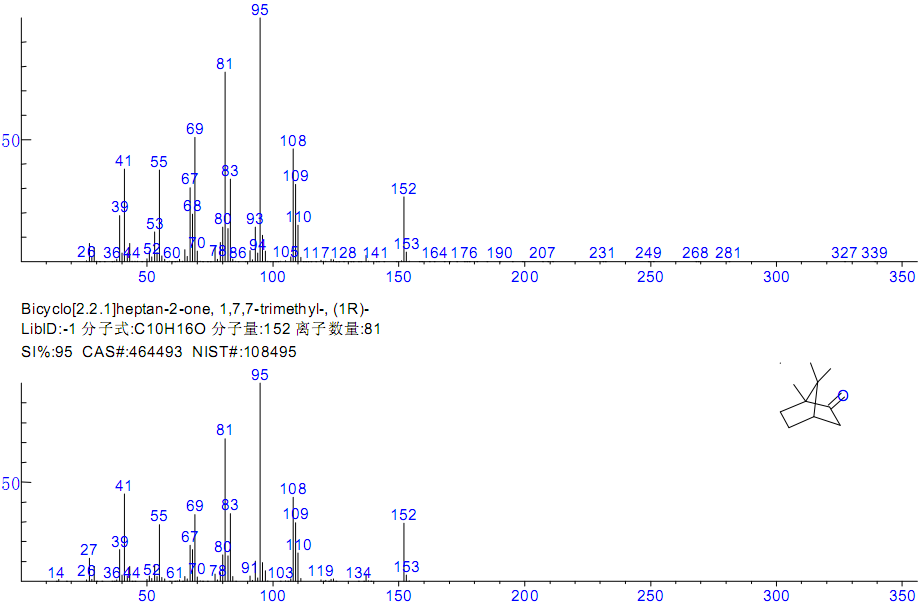
Figure 2 Mass spectra and search result of peak No. 9
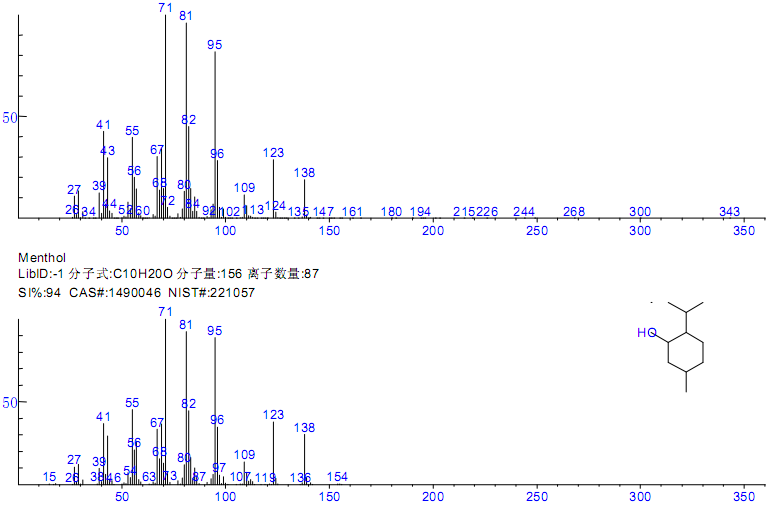
Figure 3 Mass spectra and search result of peak No. 12