Determination of Manganese, Copper, Lead, Cadmium, Silver, Cobalt, Molybdenum
1 Sample solution preparation:
49mL drinking water was measured and placed in a 50mL volumetric flask, 1.0ml nitric acid was added, to acidify water sample. Shaken, and set aside for later use .
2 Experimental equipment and reagents:
AA7000 series atomic absorption spectrophotometer (with manganese, copper, lead, cadmium, silver, cobalt, molybdenum, nickel, beryllium, chrome hollow cathode lamps, EWAI Inc.)
Nitric acid (HNO3): excellent grade purity
Manganese standard solution (National Reference Materials Research Center)
Copper standard solution (National Reference Materials Research Center)
Lead standard solution (National Reference Materials Research Center)
Cadmium standard solution (National Reference Materials Research Center)
Silver standard solution (National Reference Materials Research Center)
Cobalt standard solution (National Reference Materials Research Center)
Molybdenum standard solution (National Reference Materials Research Center)
Nickel standard solution (National Reference Materials Research Center)
Beryllium standard solution (National Reference Materials Research Center)
Chromium standard solution (National Reference Materials Research Center)
3 Instrument conditions
Mn: ashing temp 400, atomization temp 2600
Cu: ashing temp 500, atomization temp 2500
Pb: ashing temp 300, atomization temp 1600
Cd: ashing tmep 300, atomization temp 1600
Ag: ashing temp 500, atomization temp 2100
Co: ashing temp 500, atomization temp 2600
Mo: ashing temp 600, atomization temp 2600
Ni: ashing temp 600, atomization temp 2600
Be: ashing temp 1100, atomization temp 2600
Cr: ashing temp 500, atomization temp 2600
4 Standard solution preparation
When measuring Pb and Cd, injection volume is 10μL, and then 5μL of 1% ammonium dihydrogen phosphate solution was added as matrix modifier and mixed in graphite tube, to eliminate interference.
5 Standard curve
49mL drinking water was measured and placed in a 50mL volumetric flask, 1.0ml nitric acid was added, to acidify water sample. Shaken, and set aside for later use .
2 Experimental equipment and reagents:
AA7000 series atomic absorption spectrophotometer (with manganese, copper, lead, cadmium, silver, cobalt, molybdenum, nickel, beryllium, chrome hollow cathode lamps, EWAI Inc.)
Nitric acid (HNO3): excellent grade purity
Manganese standard solution (National Reference Materials Research Center)
Copper standard solution (National Reference Materials Research Center)
Lead standard solution (National Reference Materials Research Center)
Cadmium standard solution (National Reference Materials Research Center)
Silver standard solution (National Reference Materials Research Center)
Cobalt standard solution (National Reference Materials Research Center)
Molybdenum standard solution (National Reference Materials Research Center)
Nickel standard solution (National Reference Materials Research Center)
Beryllium standard solution (National Reference Materials Research Center)
Chromium standard solution (National Reference Materials Research Center)
3 Instrument conditions
Mn: ashing temp 400, atomization temp 2600
Cu: ashing temp 500, atomization temp 2500
Pb: ashing temp 300, atomization temp 1600
Cd: ashing tmep 300, atomization temp 1600
Ag: ashing temp 500, atomization temp 2100
Co: ashing temp 500, atomization temp 2600
Mo: ashing temp 600, atomization temp 2600
Ni: ashing temp 600, atomization temp 2600
Be: ashing temp 1100, atomization temp 2600
Cr: ashing temp 500, atomization temp 2600
4 Standard solution preparation
When measuring Pb and Cd, injection volume is 10μL, and then 5μL of 1% ammonium dihydrogen phosphate solution was added as matrix modifier and mixed in graphite tube, to eliminate interference.
| Element | Concentration(μg/L) | ||||
| Mn | 0 | 2.0 | 4.0 | 8.0 | 12.0 |
| Cu | 0 | 5.0 | 10.0 | 20.0 | 30.0 |
| Pb | 0 | 5.0 | 10.0 | 15.0 | 20.0 |
| Cd | 0 | 0.2 | 0.4 | 0.8 | 1.2 |
| Ag | 0 | 1.0 | 2.5 | 5.0 | |
| Co | 0 | 5.0 | 10.0 | 20.0 | 30.0 |
| Mo | 0 | 5.0 | 10.0 | 20.0 | 30.0 |
| Ni | 0 | 5.0 | 10.0 | 20.0 | 30.0 |
| Be | 0 | 0.2 | 0.6 | 1.0 | 1.4 |
| Cr | 0 | 5.0 | 10.0 | 15.0 | 20.0 |
5 Standard curve
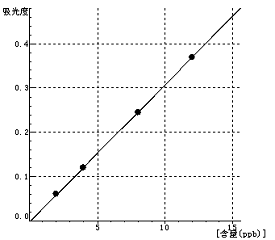 Mn Curve equation:y=0.0311*x-0.0033 Linearity coefficient:0.99997 |
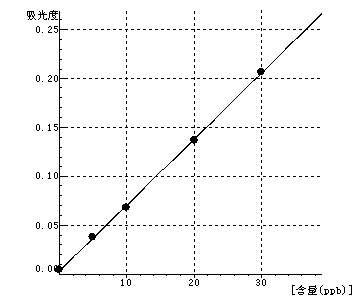 Cu Curve equation:y=0.0067*x+0.0032 Linearity coefficient:0.99975 |
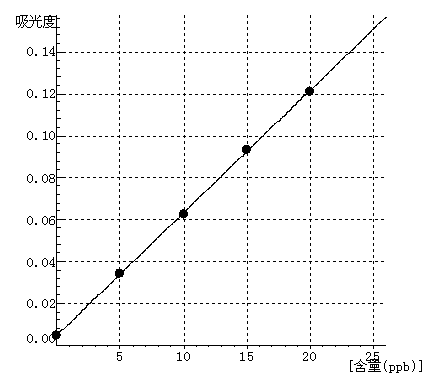 Pb Curve equation:y=0.0059*x+0.0047 Linearity coefficient:0.99992 |
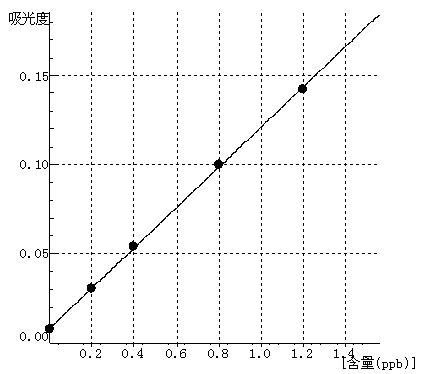 Cd Curve equation:y=0.1130*x+0.0081 Linearity coefficient:0.99985 |
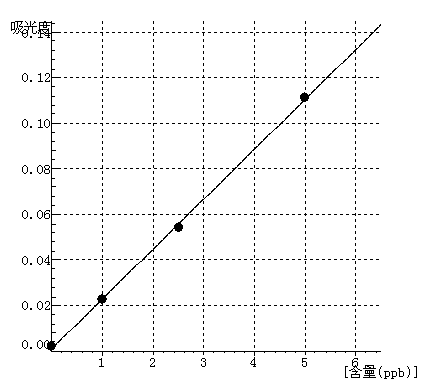 Ag Curve equation:y=0.0219*x+0.0011 Linearity coefficient:0.99960 |
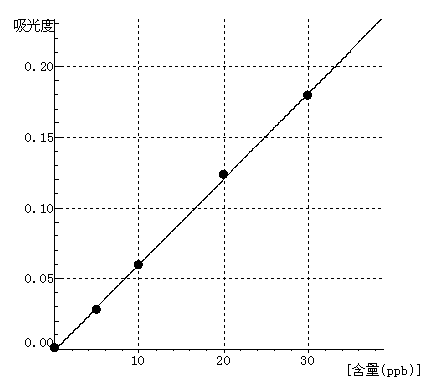 Co Curve equation:y=0.0061*x-0.0005 Linearity coefficient:0.99973 |
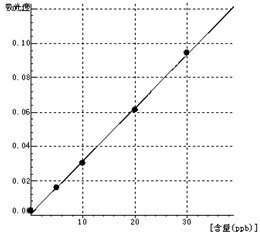 Mo Curve equation:y=0.0031*x+0.0007 Linearity coefficient:0.99930 |
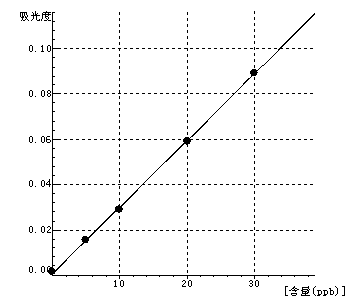 Ni Curve equation:y=0.0029*x+0.0007 Linearity coefficient:0.99982 |
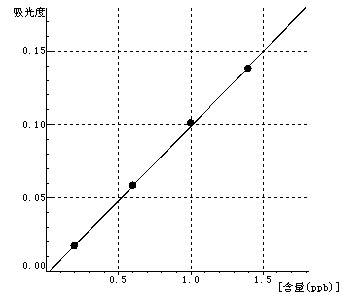 Be Curve equation:y=0.1011*x-0.0023 Linearity coefficient:0.99971 |
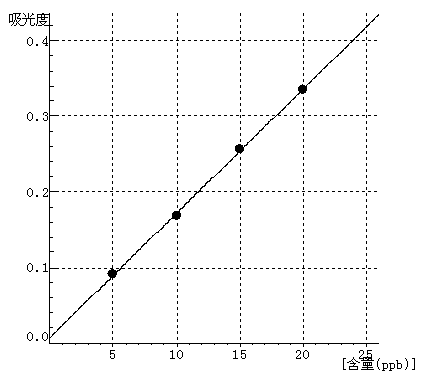 Cr Curve equation:y=0.0164*x+0.0074 Linearity coefficient:0.99962 |